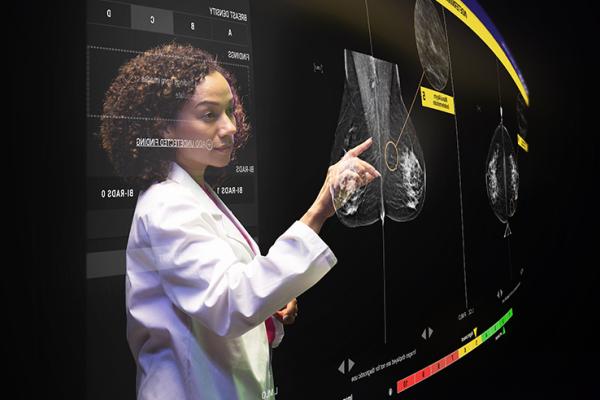
Helping Women Discover That Better Is Possible
Hologic is committed to raising awareness around conditions like fibroids and abnormal uterine bleeding and the latest technologies that can help treat them.
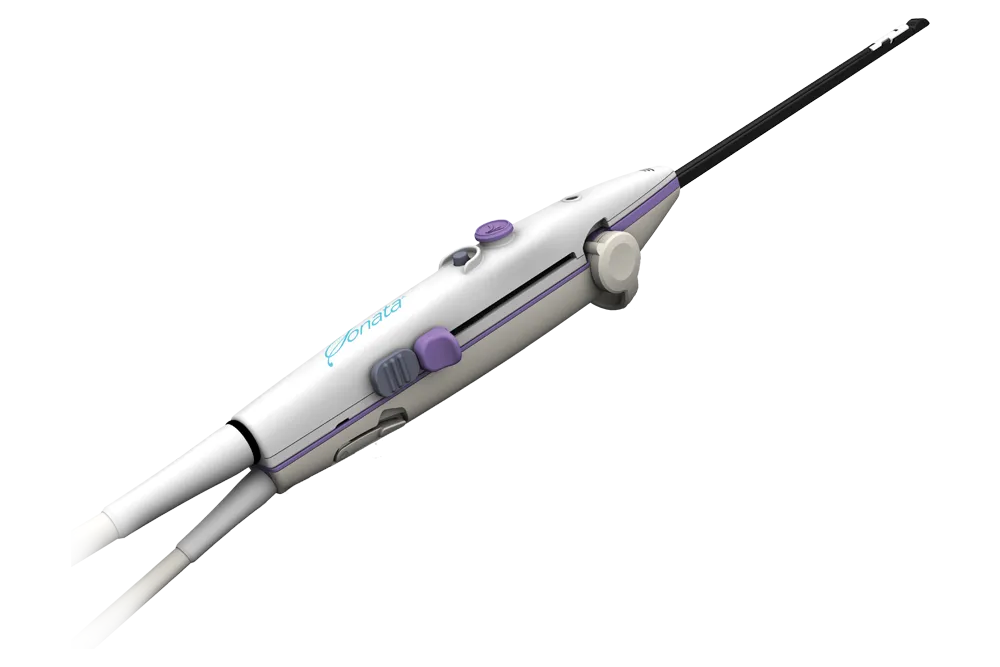
An Incisionless Solution for Fibroid Relief
Over 15,000 women have chosen the Sonata® procedure.
Support
How can we help you?
Contact Us
Find a sales rep or dealer near you.
Education
Videos, webinars and more.
Patient Materials
Why you want to ask for Hologic.
Hologic News
About Hologic
Hologic helps women around the world live healthier lives.
We develop innovative medical technologies that effectively detect, diagnose and treat health conditions and raise the standard of care globally.
We know women face unique health challenges at every stage of life, and we are passionate about being a force for good.
36
Countries
1985
Hologic is founded
4,100+
Patents
~7,000
Employees
Careers
Hologic is propelled by the passion and courage of our people to make a difference and innovate in all we do. We believe everyone should feel welcomed, valued and heard, because great ideas come from a diversity of views and experiences. We are proud to create opportunities for those who want to grow their careers while making the world a better, healthier place.