Quality Management System
The purpose of Hologic’s Quality Management System (QMS) is to consistently meet customer requirements and enhance customer satisfaction. The performance of the QMS is documented and maintained to ensure its effectiveness in accordance with applicable standards and regulatory requirements. It is aligned with Hologic’s mission and values, and constitutes a framework that ensures continuous improvement.
The Hologic QMS includes multiple regulatory certifications, such as the current ISO 13485:2016 certifications, which are held at each of our sites. In addition, each Hologic site is responsible for maintaining an adequate organizational structure to ensure that products manufactured there are designed and produced in compliance with the requirements of the QMS. Individuals responsible for performing and verifying work affecting quality are defined. These individuals have the independence and authority to assure product quality and conformance to specifications and quality objectives. Each site has an organizational chart that clearly delineates roles and functional responsibility.
The Hologic QMS takes into account requirements unique to our business and the regulatory requirements of the healthcare industry. Hologic has documented procedures for providing complaint notification to the appropriate regulatory authorities. These meet specified reporting criteria for adverse events and issuance of advisory notices. These records are maintained in accordance with Hologic’s record retention program.
Hologic's Quality Management System:
Our field action procedure defines the requirements and conditions under which a recall (field correction or removal), customer notification, safety alert, market withdrawal, and/or field safety corrective action take place.
This procedure applies to all facilities and organizations within Hologic. It applies to all products manufactured by or for us and is applicable when a Hologic product, including software, has entered commercial distribution and is suspected of posing a risk to patient health or violating applicable regulations. All Hologic entities follow the minimum requirements defined in our Corporate Standard Operating Procedures. If additional local laws, regulations and/or standards are required, Hologic sites are responsible for developing a local procedure to ensure full compliance with applicable requirements.
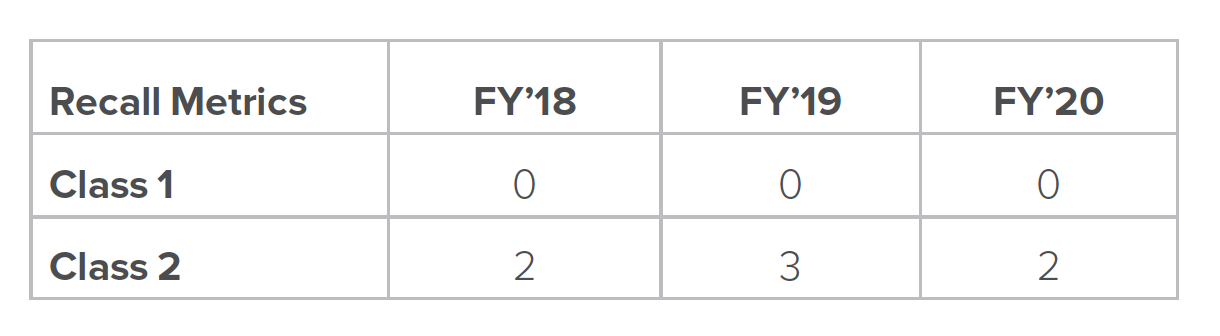
Customer satisfaction is a key priority. Our procedures establish requirements for complaint handling for marketed products in accordance with established regulations across all sites that are governed by our corporate quality system. They also allow flexibility, where appropriate, based on the nature of the products and processes at the individual sites. The requirements of these procedures apply to all finished products manufactured or distributed by Hologic.
Our quality audit procedure provides parameters for our internal audit program. Internal audits are conducted to determine whether our Quality Management System at all global sites and regions complies with applicable internal and external requirements, ensuring that these are effectively implemented and maintained.
All Hologic employees are required to complete training in Quality System requirements and to acknowledge and adhere to our Hologic Quality Policy.
Hologic requires a process and system to show compliance to chemical regulations where Hologic product is marketed and sold. In order to comply with applicable chemical regulations, Hologic has implemented a process and system to ensure all Substances controlled by chemical regulations within Hologic product are identified, monitored and reported as applicable.