Leadership Message
For nearly four decades, Hologic has championed greater health and well-being for all women — no matter where they live, how much money they make or what their education is — because we believe that women are the cornerstones of society and improving women’s health has a powerful ripple effect on families and communities. We firmly believe that our success as a company is fundamentally tied to our ability and commitment to help millions of women worldwide live healthier lives. Because of this belief, we focus on developing industry-leading innovative technologies that enable early detection, diagnosis and treatment of life-changing health conditions that affect women. This is our reason for being.
Stephen P. MacMillan

About Hologic
Hologic helps women around the world live healthier lives. We develop life-changing medical technologies that deliver exceptional results essential to effectively detect, diagnose and treat health conditions that affect women globally.

A Year in Review
Employee engagement drives our lifesaving innovations, informs our environmental progress and inspires our community-based giving. See Page 4
Business Overview
Hologic’s core strengths in women’s health create the foundation for diversified, durable growth and dependable performance. See Page 5Sustainability Strategy
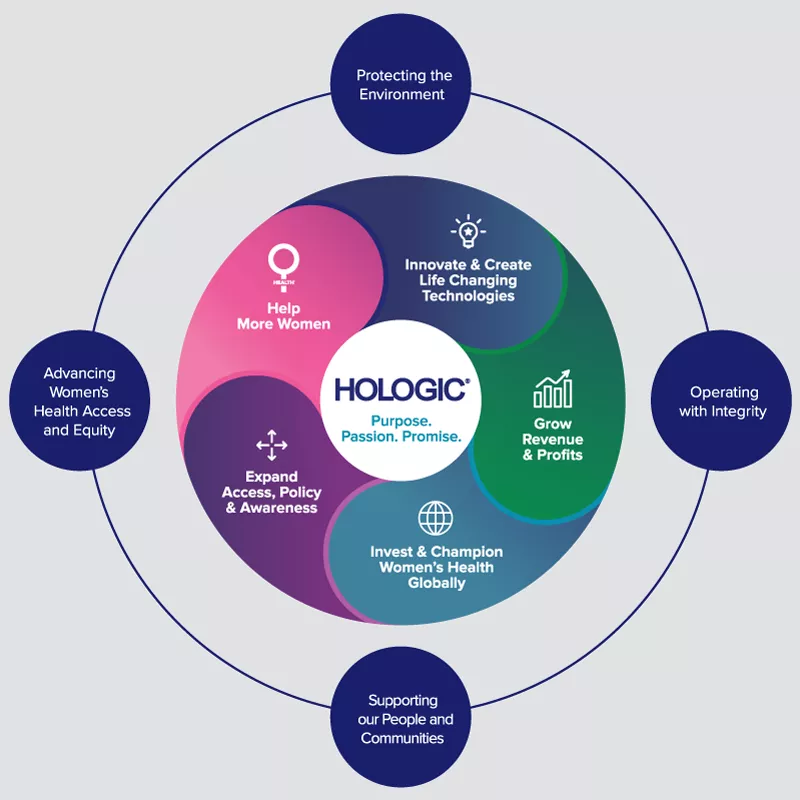
At Hologic, women’s health is more than a business, it is our purpose. As a science-based company, Hologic has a long-standing approach to operate responsibly and sustainably with unwavering dedication to product quality, clinical differentiation, customer relationships, remarkable talent, engagement, community investment and environmental stewardship. With sustainability embedded in our business, we are putting our corporate objectives and our purpose in action.
Protecting the Environment
We are committed to respecting the inextricable connection between the health of the planet and the health of our patients and communities.

Environment: Our Strategy
We recognize the inextricable connection between the health of the planet and the health of our patients and communities. See Page 29
Drive Climate Action
Climate action is connected to our purpose to enable healthier lives everywhere, every day and our approach to operating responsibly. See Page 30Advancing Access and Equity in Women’s Health
We are committed to greater health and well-being for all women, their families and their communities. We are committed to advancing the highest standard of science — and providing every woman access to the science we develop.

Access: Our Strategy
We are committed to advancing the highest standard of science — and providing every woman access to the science we develop. See Page 12
Drive Innovation and Affordability
Our life-changing advances in breast, cervical, gynecologic, skeletal and sexual health during the past four decades are rooted in science and clinically proven to deliver greater certainty and peace of mind for patients everywhere. See Page 13
Promote Partnerships
We champion women’s health globally, working in partnership with others to raise awareness, inspire policy and take action to break down barriers that affect women disproportionately and prevent them from receiving the care they need and deserve. We are a catalyst for change in the world. See Page 18Supporting Our People and Communities
We are committed to developing our people in a culture where everyone has the opportunity to thrive and contribute to a shared purpose: enabling healthier lives everywhere, every day.

People: Our Strategy
We are committed to developing people in a culture where everyone has the opportunity to thrive and contribute to a shared purpose: enabling healthier lives everywhere, every day. See Page 49
Curating an Inclusive, Engaged and Innovative Culture
We believe everyone should feel welcome, heard and valued because great ideas come from a diversity of views and experiences. See Page 51Operating with Integrity
We are committed to putting our purpose, passion and promise into action while maintaining the highest level of integrity and ethical behavior in our business operations.