Breast Imaging
Leading the way in breast imaging. Hologic's breast imaging technology includes state-of-the-art mammography systems and advanced AI and software solutions for sophisticated breast image analysis.
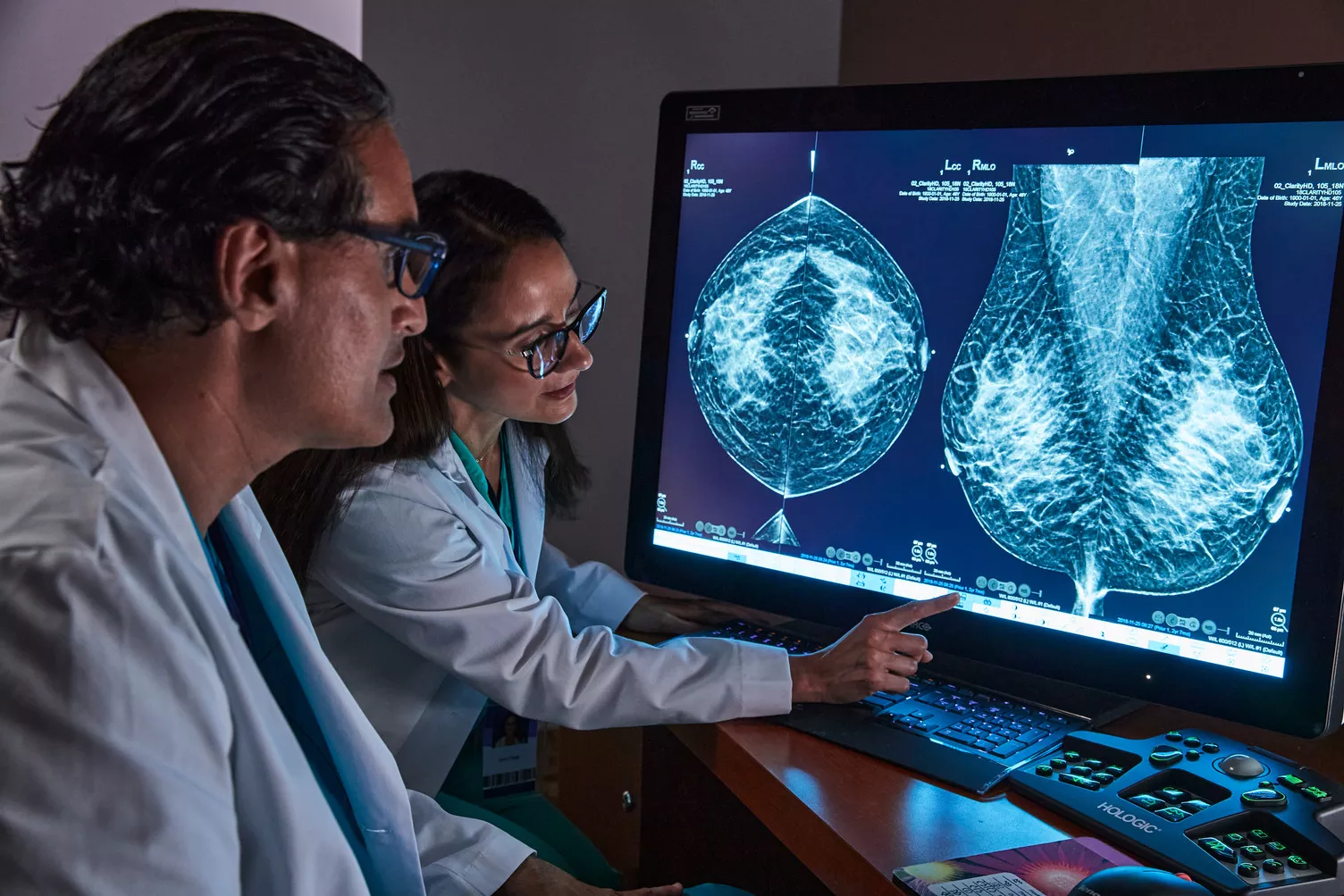
Advance Early Detection & Diagnostic Accuracy
Our integrated breast imaging technologies are designed for superior breast cancer screening1, from mammography systems that prioritize patient comfort and provide workflow enhancements, to leading-edge AI software and modern workstations for proactive and decisive reading.
Cutting-Edge Imaging
Confident Cancer Detection
Compassionate Interventional Solutions
Diagnostic & Data Efficiency
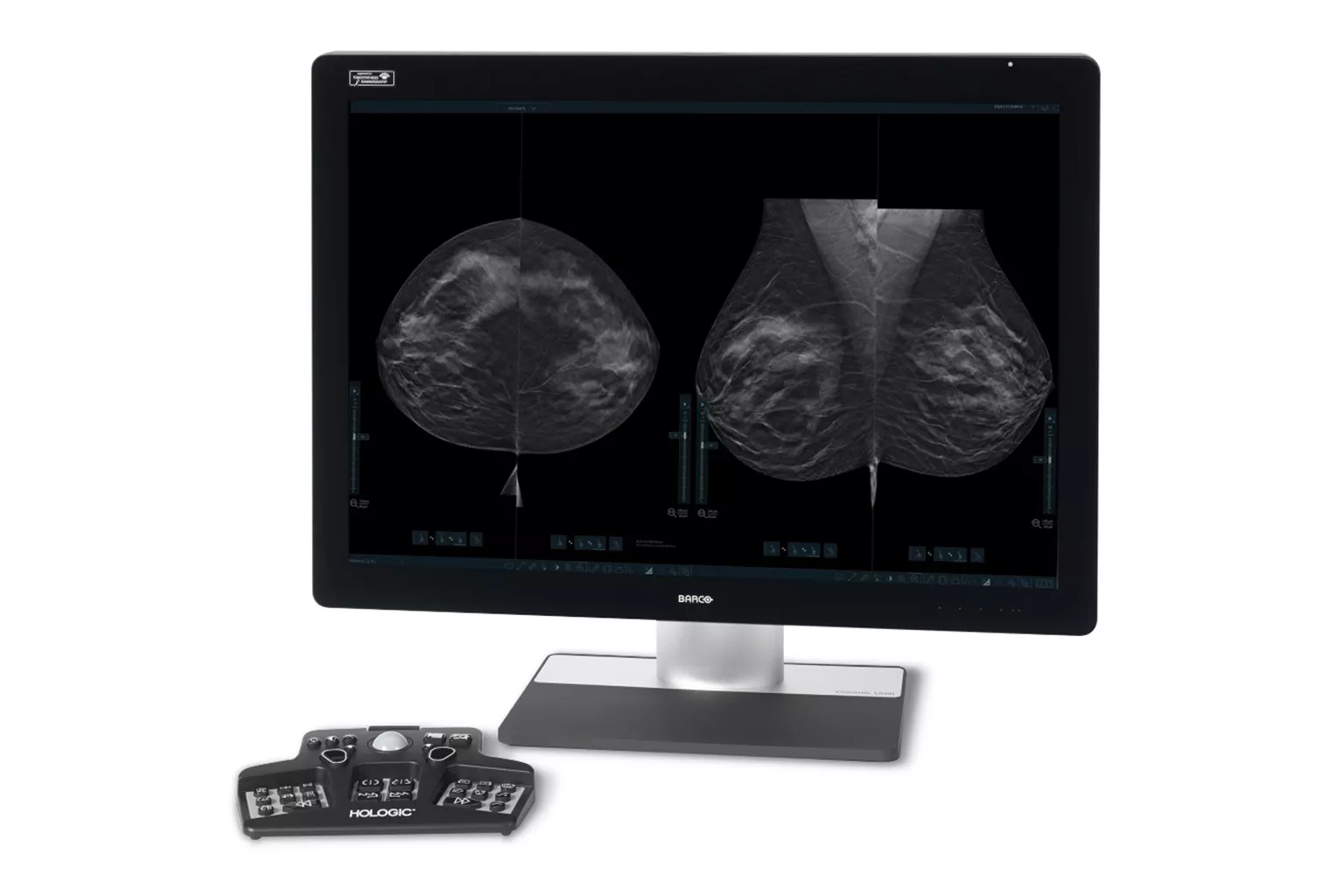
Mammography
Exceptional image quality is important to us. It has inspired our goal to improve diagnostic clarity and the vision to identify cancers quickly and accurately, even in dense breasts. All of our systems are built with patient compassion in mind, from shorter compression times to features designed to maximize comfort.2,4

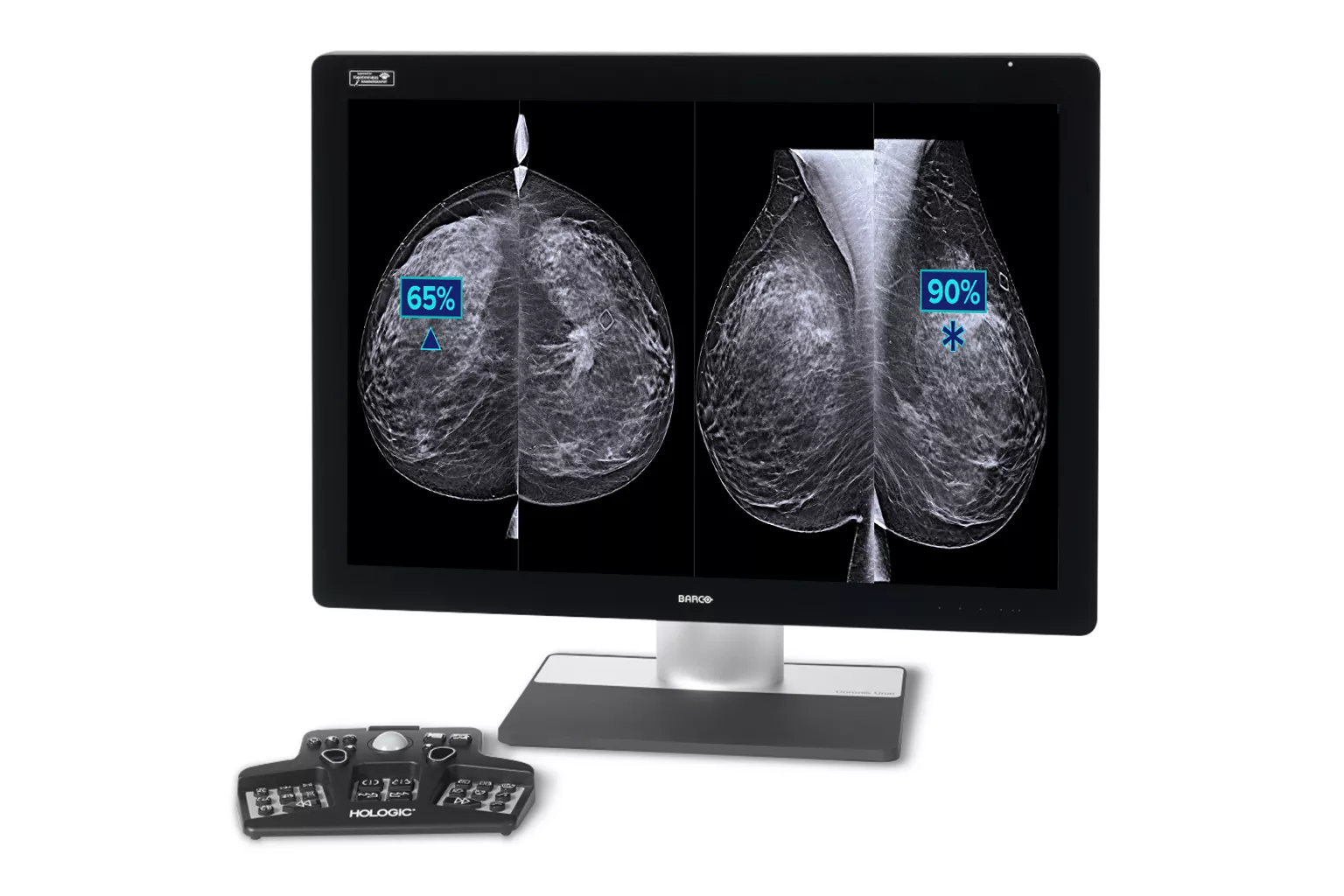
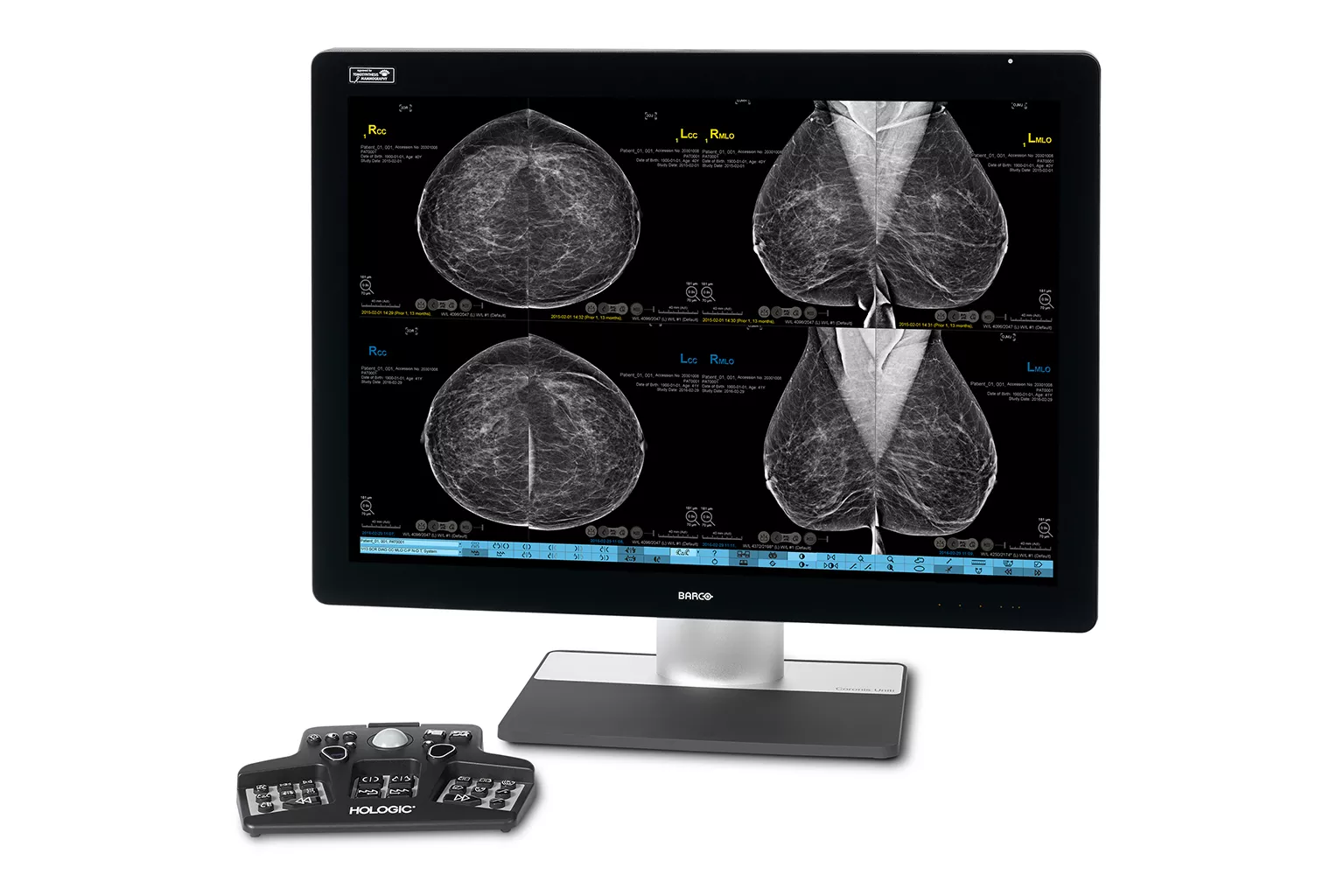
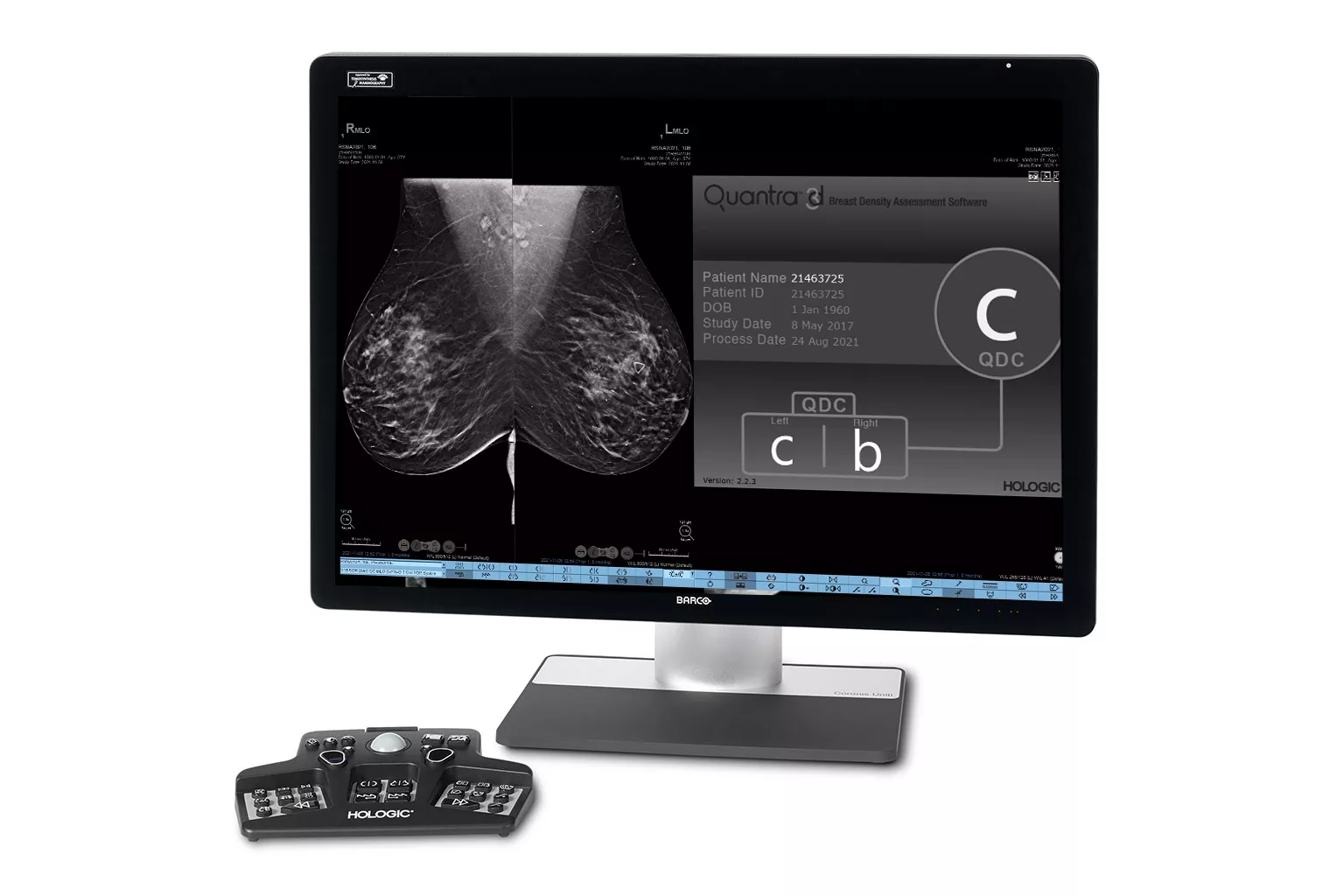
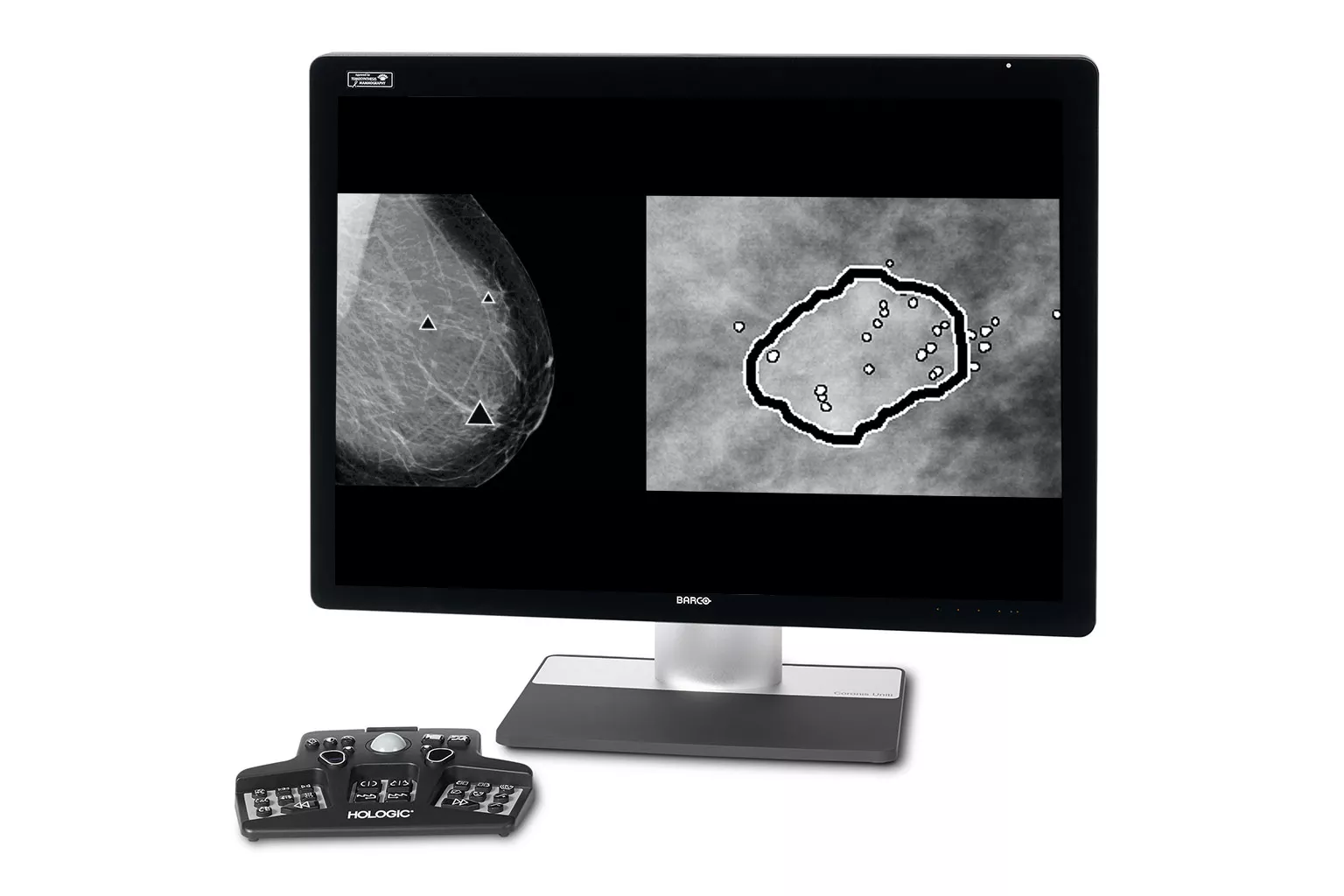
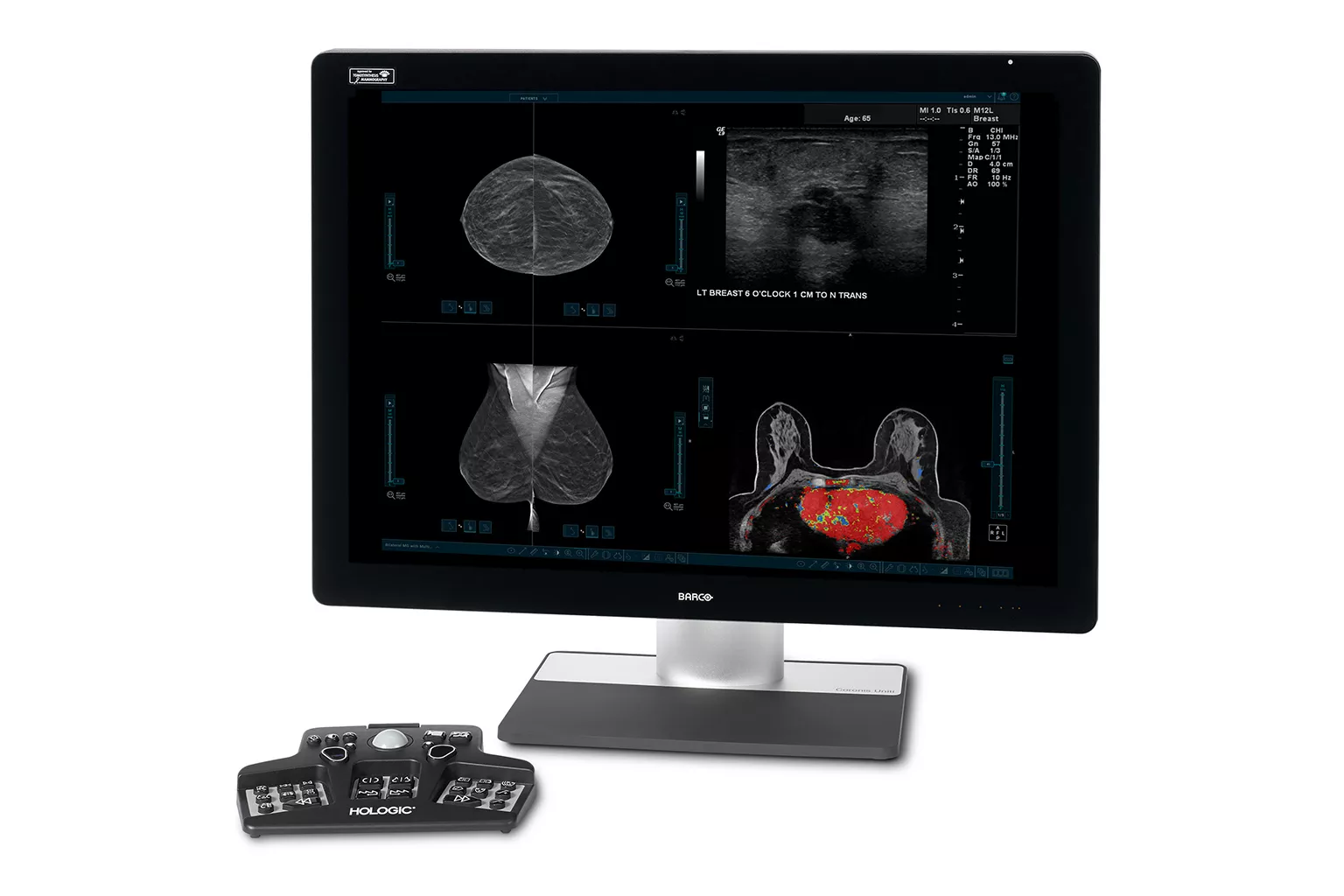
Image Analysis & AI
Our breast image analysis technology is designed with cutting-edge science and powerful algorithms to accelerate discovery of suspicious lesions, improve reading efficiency and continually sharpen image reading precision.8
Decades of Continuous Innovation
Our pioneering breast health technology supports best-in-class detection and diagnosis, engineered for speed and accuracy.1
3.7s
Scan time – the industry’s fastest tomosynthesis scan2 on the 3Dimensions™ Digital Mammography System.
1 hour
Daily saving in image interpretation time3,7,8 with 3DQuorum® Imaging Technology.
Additional cancer
+9% improvement in observed reading for approximately 1 additional cancer found for every 10 identified by a radiologist5.
70 micron
Hologic Clarity HD® imaging technology provides the highest resolution 3D images in the industry6 to clearly see subtle lesions and fine calcifications.
Visit Our Virtual Hospital
Browse our portfolio of Breast Health solutions in 3D. See how you can unlock the advantage of time across the entire Breast Continuum of Care.

1. Compared to 2D alone. FDA PMA Approval P080003 2. Data on file DHM-05051_002 3. Report: CSR-00116, based on 8 hours of image interpretation time per day. 4. Smith, A. Improving Patient Comfort in Mammography. Hologic WP-00119 Rev 003 (2017) 5. U.S. Food and Drug Administration, Center for Device and Radiological Health. (2020, November 18). Genius AI Detection K201019 510(k) Summary. IN1FDA Clearance: K201019 *Based on analyses that do not control type I error and therefore cannot be generalized to specific comparisons outside this particular study. In this study: The average observed AUC was 0.825 (95% CI: 0.783, 0.867) with CAD and 0.794 (95% CI: 0.748, 0.840) without CAD. The difference in observed AUC was +0.031 (95% CI: 0.012, 0.051). The average observed reader sensitivity for cancer cases was 75.9% with CAD and 66.8% without CAD. The difference in observed sensitivity was +9.0% (99% CI: 6.0%, 12.1%). The average observed recall rate for non-cancer cases was 25.8% with CAD and 23.4% without CAD. The observed difference in negative recall rate was +2.4% (99% CI: 0.7%, 4.2%). The average observed case read-time was 52.0s with CAD and 46.3s without CAD. The observed difference in read-time was 5.7s (95% CI: 4.9s to 6.4s). www.accessdata.fda.gov/cdrh_docs/pdf20/K201019.pdf. FDA clearance K221449, K230096 6. P080003/S006 7. Physician Labeling: MAN-06153 8. Based on 8 hours of image interpretation time per day, compared to radiologists not using not using 3DQuorum, FDA approval number.P080003/S008
Related Portfolio & Solutions
Breast Health Continuum of Care
Time is precious when it comes to effective detection, diagnosis and treatment of breast cancer. We strive to save you time at every step along the Breast Health Continuum of Care ensuring more women have more time in better health.
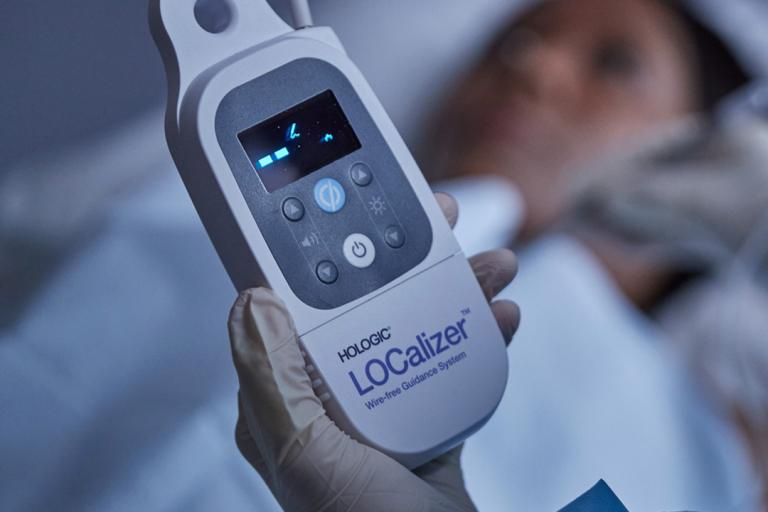
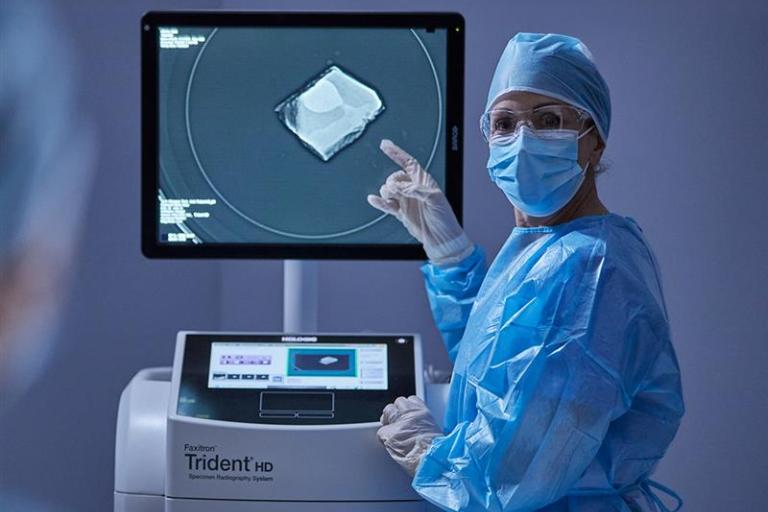
Breast Surgery Solution
Explore breast cancer surgery products from Hologic, including localization systems and wires, surgical markers and specimen imaging systems. The future of breast surgery is here.

Breast Biopsy Solution
Explore Hologic's innovative solutions for breast biopsy procedures, designed for more reliable diagnosis and improved patient care. Learn about our cutting-edge technologies and products.

AI Software Solutions
We are pioneers in using AI to drive accuracy and efficiency for breast cancer detection. Our world-class AI portfolio includes evidence-backed solutions for powerful breast imaging, reading analysis, and reporting assistance.